Contract Research – From Concept to Commercialisation
Poulpharm conducts GLP and non-GLP studies for the development and validation of animal-health products. Our dedicated research team partners with pharmaceutical, nutraceutical and feed companies to deliver reliable, regulatory-compliant data that supports product registration and market launch.
Our Contract Research Services Include
Efficacy and safety trials in different spieces
(bacteria, virus, parasites)
Feed additive evaluation
(digestibility, safety, performance)
Vaccine testing and immunological studies
Product registration support
(data, documentation, reporting)
What Sets Us Apart
Study designe customised to your product, region and regulatory requirements
Sample collection and data management done in-house
High standard of veterinary oversight and laboratory integration
Transparency, clear reporting and timely delivery
Contract research
As an independent contract research partner for animal health research in poultry, rabbits and pigs, we specialize in customer-oriented solutions for all research and drug development needs, ensuring quality, efficiency, cost-effectiveness and scientific integrity. To this end, we have an extensive team of experienced Study Directors, researchers, veterinarians, laboratory technicians and animal care staff who work together as a team to bring your study to a successful conclusion.
As a CRO we assist our clients along the pivotal steps to register and commercialize their products, whether these are veterinary medicine, feed additives or feed products.
During development, a Proof Of Concept study can reveal the potential of your product. This might range from straightforward efficacy to evaluation of underlying mechanisms of action. Our experts in the various fields will readily assist you in setting up the optimal study model.
In a follow-up step, extensive registration trials will be performed to demonstrate your product’s efficacy and safety. These trials will be conducted by a team of Study Directors, lab technicians and animals care personnel trained on the necessary requirements according to VICH and OECD guidelines. To complete this process successfully, our regulatory department can further support you on regulatory requirements to enter the market.
Finally, we have the necessary experience and expertise to assist you in marketing studies to promote your product. To this end, next to the controlled (laboratory) trials at our facilities, we also offer field trials. In these latter, your product can be tested at large scale, offering field information, user experience and an opportunity to penetrate the market.
Depending on the aim of the trial, Poulpharm comprises several sites to conduct poultry, rabbit and pig trials. As such we have the flexibility to vary from small scale to large scale in vivo studies.
Facilities include:
- Floorpens (Poultry, rabbits and pigs)
- Battery cages (Poultry for fattening and laying hens)
- Pig boxes (Currently <50kg/animals, near future also >50kg/animal)
- SUIT cages (Systems to Ultimately Isolate Threats, Poultry)
- BSL2 and BSL3 level isolators (Poultry)
- HEPA-filtered isolator rooms (40-60m² to accommodate floorpens or pig boxes)
- Compartments for field trials (4 compartments for Poultry)
- Fattening pig houses (2)
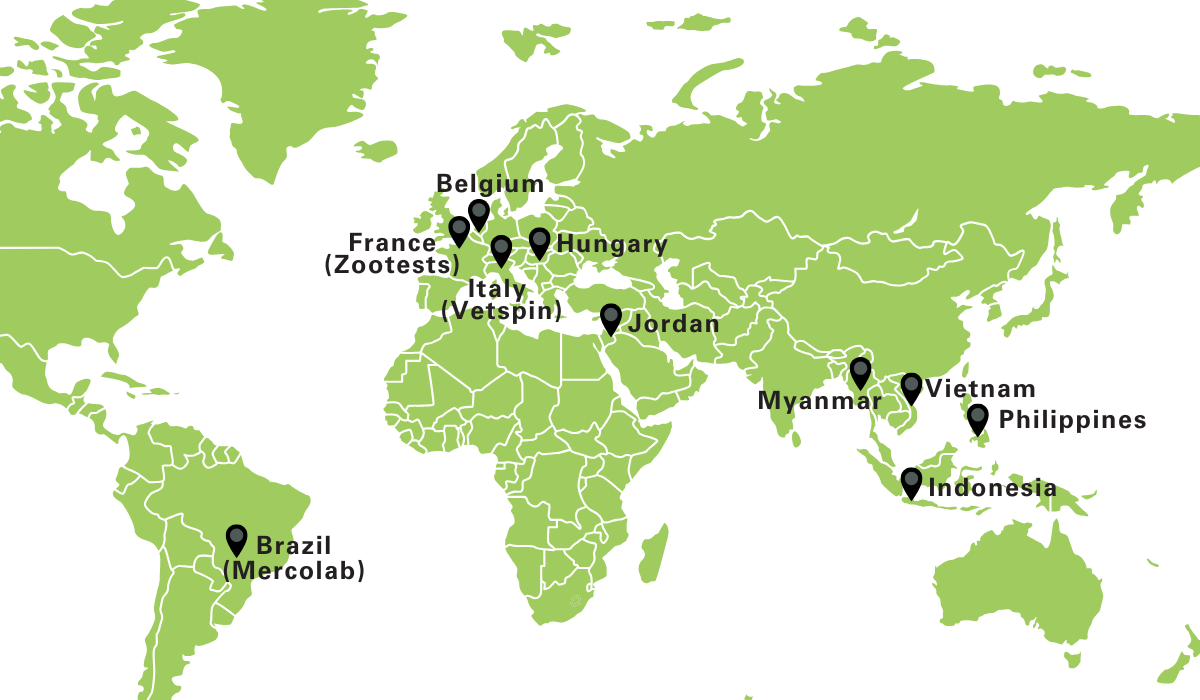
Moreover, via an international network of dedicated farms and with access to an extensive veterinarian network we can offer real-life field trials for your demand. This network includes farms and veterinarians in different EU countries as well as Asia and Latin America (Belgium, France, The Netherlands, Portugal, Italy, Hungary, Vietnam, Philipppines, Brasil, …).
Poulpharm offers a wide range of models whether your products are veterinary medicine or vaccines, feed additives, feed products or biocides:
- Efficacy
- Dose-determination
- Target Animal Safety
- Compatibility
- Toxicity
- Tissue Residue
- Pharmacokinetics
With a team of Scientific experts in different fields and an extensive biobank, we can offer infection models with following pathogens:
- Parasites
- Coccidiosis
- Histomoniasis
- Helminth infections
- Red blood mites
- …
- Bacteria
- Necrotic enteritis
- Bacterial enteritis
- E.coli
- Salmonellosis
- Mycoplasma
- Gallibacterium
- Enterococcus
- Streptococcus
- Staphylococcus
- …
- Viruses
- Infectious Laryngotracheitis
- Infectious Bronchitis
- Marek’s disease
- Adenovirus
- Reovirus
- Fowl-pox
- Avian Metapneumovirus infection
- Avian encephalomyelitis
- Swine Influenza Virus
- Porcine Reproductive Respiratory Syndrome Virus
- …
Furthermore, our non-infection models include
- Performance
- Lipopolysaccharide (LPS)
- Footpad dermatitis and lameness
- Leaky gut
- Heat stress
- Mycotoxin
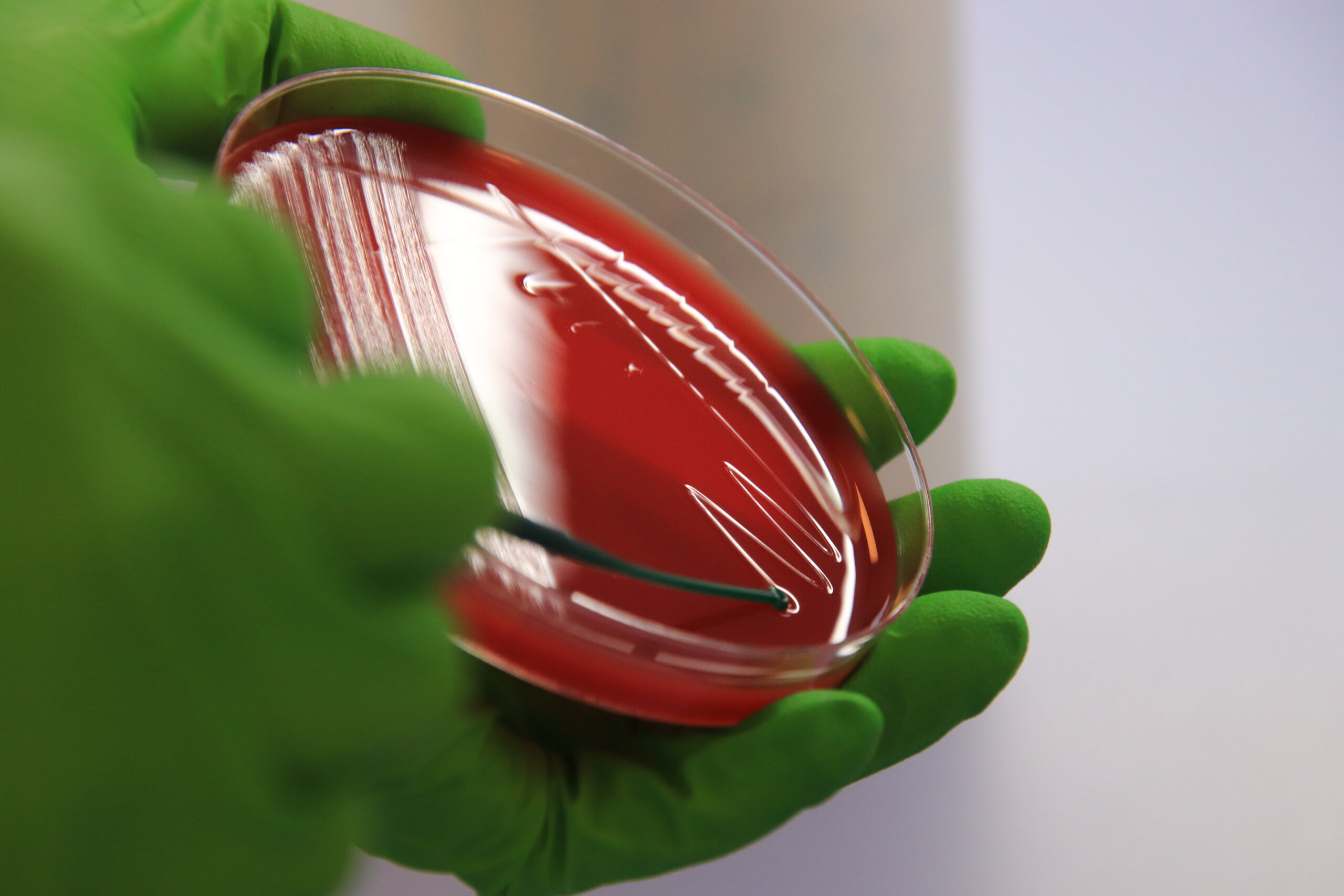
Samples collected during in vivo trials, will either be processed in one of our own laboratories with highly-trained personnel and specialized equipment
Or assessed by a partners from our wide network of external collaborators.
Our Services
read service FAQ’s
Tel: +84 707 465 167
E-mail: info.vn@poulpharm.com
Address: Đ/c: L17-11, Tầng 17, Tòa nhà Vincom Đồng Khởi, 72 Lê Thánh Tôn, Phường Bến Nghé, Quận 1Thành phố Hồ Chí Minh – Việt Nam
VAT 0316330244
Tel: +63 966 452 5559
E-mail:info.ph@poulpharm.com
Address: Kalaw Compound, Brgy. Batong Malake, Los Baños, Laguna, Philippines
Tel: +91 757 917 6100
E-mail: info.mm@poulpharm.com
Address: No. R8 (C+D), Khayae Pin Yate Mon Housing 2, Mingalardon Township,11021, Yangon – Myanmar
Tel: +91 757 917 6100
E-mail: info.id@poulpharm.com
Address: Alamanda Tower, 19th Floor Unit G, Jalan TB Simatupang Kaveling 22-26, South Jakarta – Indonesia
Tel: +36 70 433 0133
E-mail: info.hu@poulpharm.com
Address: 1183, Budapest Csaba utca 30. – Hungary
VAT HU27074402