Advancing Poultry Health Through Science & Collaboration

Our Facilities & Quality
At our headquarters in Izegem (Belgium) and affiliated sites globally, our laboratories are equipped for bacteriology, virology, molecular diagnostics, serology, histology and controlled-environment animal studies. We adhere to strict biosecurity and quality standards, including ISO 17025 accreditation and GLP/GCP compliance for research projects.
Quality control is built into every step—from sample intake and data management to reporting and client follow-up.
SAS-Global Connection:
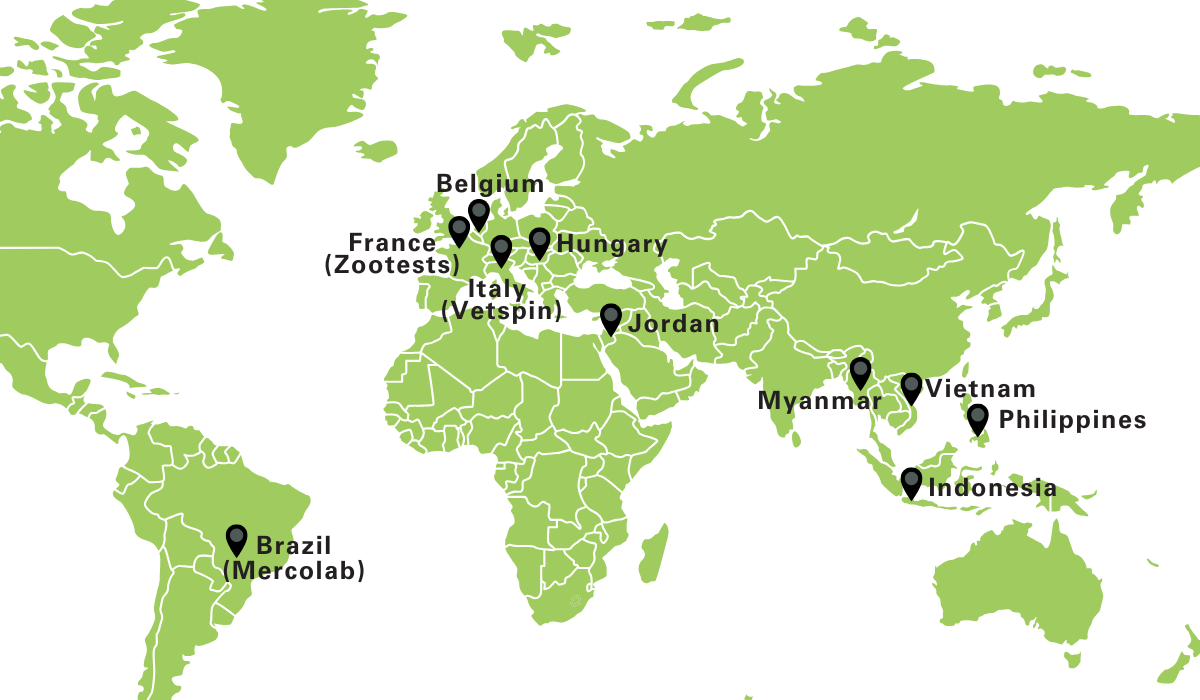
Poulpharm is part of the SAS-Global group an umbrella brand uniting leading animal health science partners. This network enhances our global reach, resource access and multidisciplinary strength, while keeping our poultry-specific expertise front and centre.

network & partners
Poulpharm owns full-service laboratories for a range of viral, bacterial and parasitic tests. The lab is ISO 17025 accredited (BELAC 610-TEST) and GLP certified. GLP compliant analytical tests for pharmacokinetic and residue studies are offered in collaboration with Vetspin (Italian satellite laboratory) or the Laboratory of Pharmacology and Toxicology, Faculty of Veterinary Medicine, Ghent University.
Development of new study models and fundamental research in collaboration with Ghent University (Chair of Health Sciences Poultry).
Finally, Poulpharm works in close collaboration with the poultry and pig industry (farmers, veterinarians, feed mills, hatcheries,…), by giving them an insight into economically important field situations.