Who we are
Poulpharm is an independent Contract Research Organization and diagnostic partner for the veterinary pharmaceutical and animal nutrition industries, and part of the SAS Global Group.
Poulpharm was founded in 2007 in Izegem, Belgium. Since then, we have been providing high-quality diagnostics and species-specific in vivo research across poultry, pigs, cattle, companion animals, hobby birds, and aquaculture. Our studies support the development and evaluation of feed additives, veterinary products, and nutritional solutions under real-life production conditions.
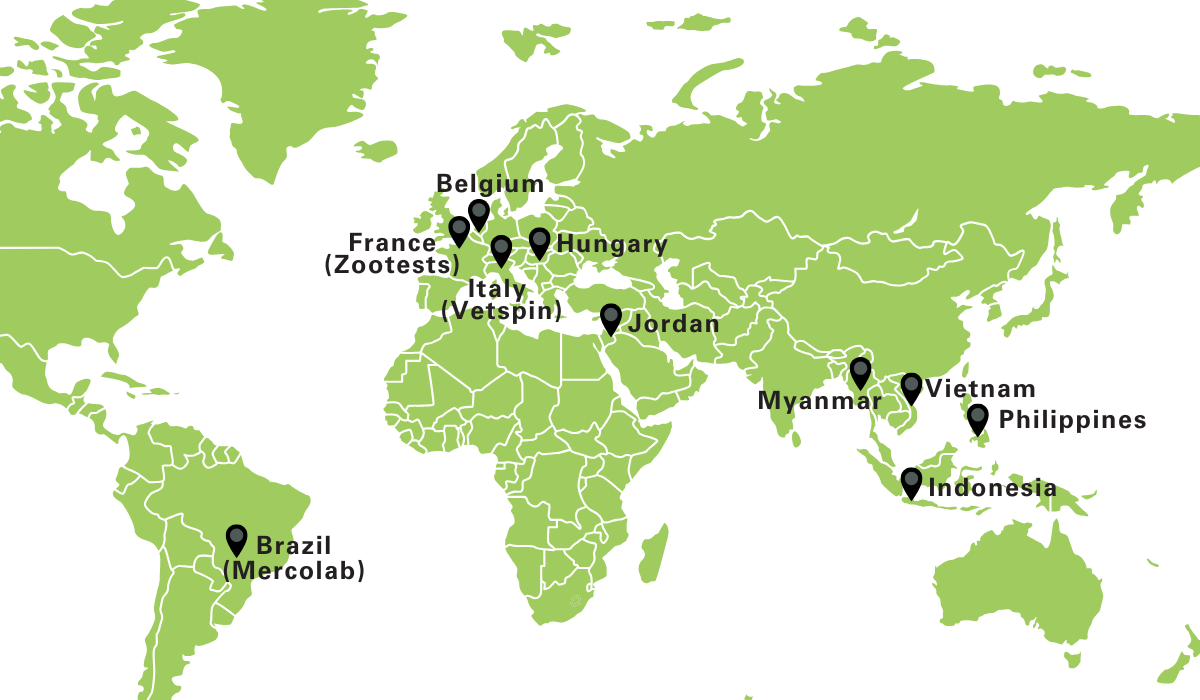
With facilities across Europe, Asia, and Latin America, we operate under GLP, GCP, and ISO 17025 standards, ensuring reliable and compliant results from diagnostics to preclinical and clinical trials.
By combining scientific expertise, practical trial design, and a global network, Poulpharm supports innovation from concept to market in animal health.

Our Mission
Over two decades of experience in veterinary diagnostics, microbiology, and immunology have shown us that modern animal health goes beyond reactive treatment – it requires integrated research, field data, and preventive solutions.
Poulpharm was created to bring veterinary science, high-quality laboratory services, and tailored research together under one roof. As part of SAS-Global, we combine specialist poultry expertise with a global network, enabling stronger collaboration, broader insights, and greater impact
Our Team
We are a multidisciplinary team of veterinarians, microbiologists, immunologists and research scientists.
All driven by a common goal: turning data into actionable insights for poultry farms, feed companies and laboratories. Each project is backed by scientific rigour and dedicated client communication.


Our Facilities & Quality
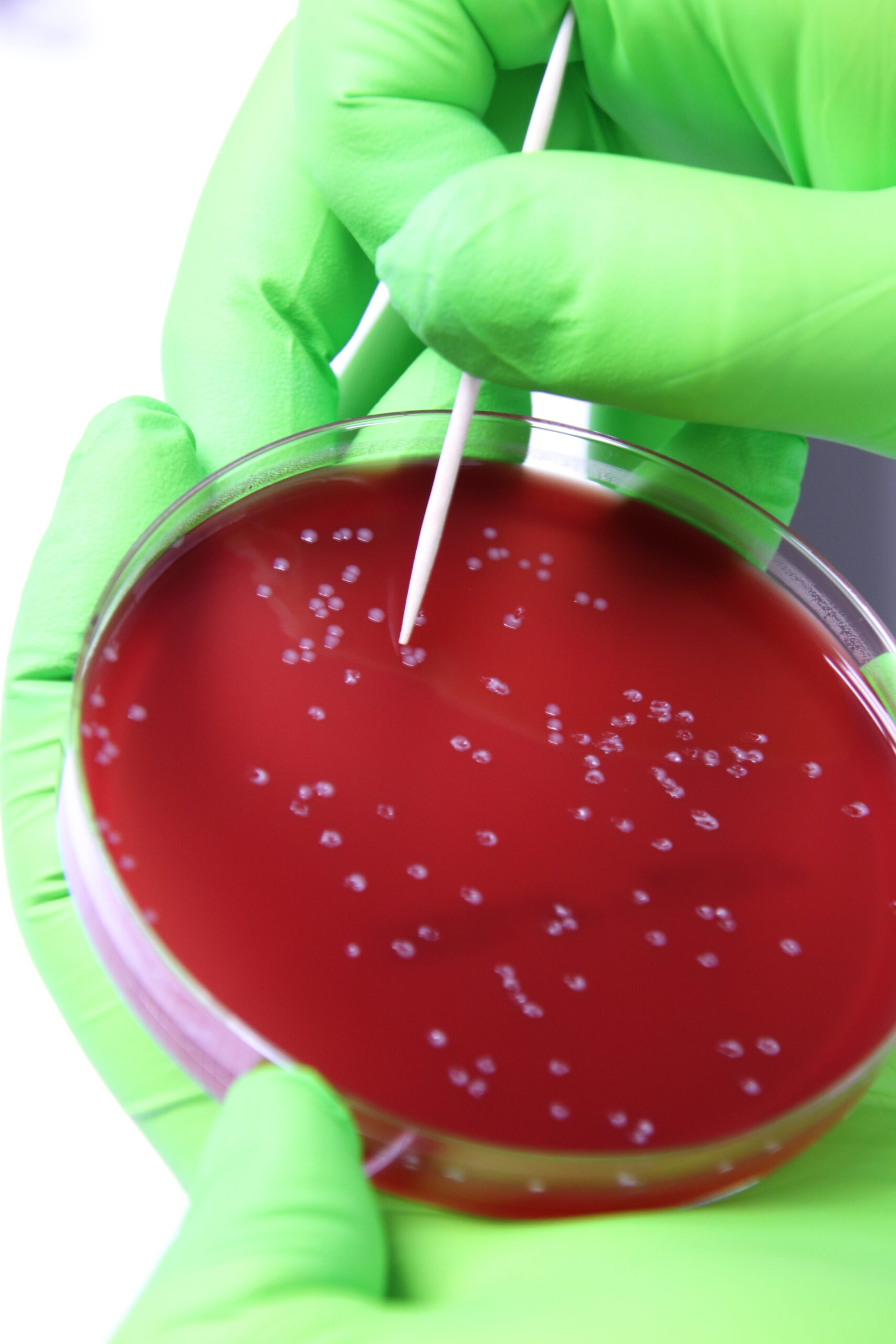
At our headquarters in Belgium and affiliated sites globally, our laboratories are equipped for bacteriology, virology, molecular diagnostics, serology, histology and controlled-environment animal studies.
We adhere to strict biosecurity and quality standards, including ISO 17025 accreditation and GLP/GCP compliance for research projects. Quality control is built into every step—from sample intake and data management to reporting and client follow-up.